ME Assistant Professor Ashish Manohar shares how mechanical engineers can contribute to improving medical imaging for better health outcomes.
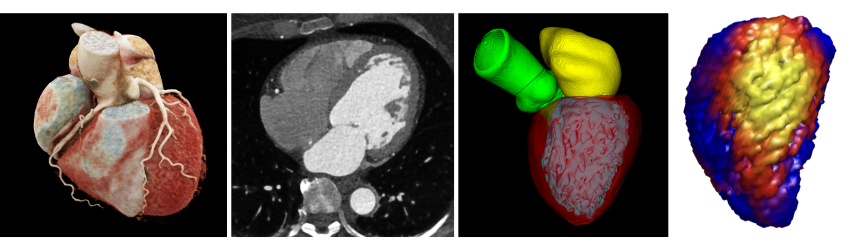
Starting with high-resolution CT scans (second image), a personalized computational modeling pipeline segments anatomical features and generates detailed 3D organ models (third and fourth images) that quantify structural and functional properties. This example shows the approach applied to the heart (first image: a visualization of a human heart CT scan). Images provided by Manohar.
When one thinks of mechanical engineering research, medical imaging may not be the first area that comes to mind, compared to energy, robotics, aerospace and more. However, UW ME has been pioneering health innovations for decades and has deep faculty expertise in medical imaging and image-based modeling.
To learn more, ME spoke to Assistant Professor Ashish Manohar, who has a specific focus in medical imaging physics. Manohar joined the department in winter 2026 and holds an adjunct appointment in radiology. His goal is to develop advanced imaging methods for computed tomography (CT), an x-ray imaging procedure that provides detailed 4D (3D with the added dimension of time) images, to improve disease diagnosis and management, particularly cardiovascular disease.

ME Assistant Professor Ashish Manohar
What is medical imaging, and why is it important in research?
Medical imaging allows us to see inside the human body non-invasively, providing critical information about anatomy, function and disease that would otherwise be impossible to obtain. With advancements in computational modeling, medical imaging goes beyond just diagnosis. Images of patients are now used as the foundation for developing patient-specific models that can guide treatment decisions, predict patient outcomes and develop personalized therapies. Put simply, medical imaging is pivotal in helping us move towards a more quantitative and personalized field of medicine.
However, the accuracy of these patient-specific models depends critically on image quality. If images are inadequate due to poor quality or artifacts, which are errors in the images, the robustness of our modeling is compromised. This is why medical imaging is both an exciting and essential area of research, where advances in image quality directly translate to more accurate patient-specific models.
What do mechanical engineers bring to the area of study?
Mechanical engineers bring a powerful combination of skills to medical imaging. We're trained in both physics and computational modeling, and those two things work together beautifully in this field. On the physics side, we understand the fundamental principles behind how imaging systems work, including signal processing, which helps us tackle challenges like reducing artifacts, improving image quality and minimizing radiation dose. On the modeling side, we know how to take those images and build computational models that can simulate what’s actually happening in a patient's body, whether that's how blood flows through an artery or how the heart contracts during each beat. So mechanical engineers end up sitting at this really interesting intersection: we work on making better images and we figure out how to extract meaningful information from those images to help patients.
UW ME is a prime example of this. Faculty like Juan Carlos del Álamo, Alberto Aliseda, Mehmet Kurt and Eric Seibel are pioneers in utilizing medical imaging to develop robust computational models for understanding human health and disease. I’m very excited to be part of this department and continue developing robust imaging methods.
How did you become interested in medical imaging?
I used to think mechanical engineering was all about turbines, airplanes, cars and energy. Originally, I wanted to study experimental fluid dynamics as it relates to aerospace. When I joined University of California San Diego (UCSD) as a master’s student, I came across Juan Carlos's work and learned how he applied fundamental fluid mechanical principles to model blood flow in the heart, and it blew my mind. I was really interested in the health applications, like being able to predict stroke risk.
While starting to work in this area, I learned that my models could only be as accurate as the images themselves. One thing led to another, and I started getting more interested in how we could develop methods to make the images themselves better. Luckily, Elliot McVeigh, a cardiac imaging expert and professor at UCSD, was looking for a Ph.D. student, and that's how I found my way into medical imaging research.
What are some of your research interests?
My work sits at the intersection of imaging physics and computational modeling. I focus primarily on cardiac CT imaging because it gives us these incredibly detailed images of the heart. You can see the complex 3D texture of the heart muscle and how the entire heart beats, all captured in a single heartbeat, which is truly amazing! The challenge, however, is figuring out how to make those images even better and extract as much useful information from them as possible.
There are also some exciting new directions I'm exploring, like improving imaging methods for pediatric patients with congenital heart disease, and figuring out how to capture high-quality images during stress testing when the heart is beating really fast. The beauty of being in mechanical engineering is that we can tackle both sides of the problem, making the images better and building better models from those images.
Tell us about your lab in ME.
My lab’s focus is in three main areas. First is imaging physics: understanding how CT images are formed and developing patient-specific approaches to optimize image quality while minimizing radiation dose (because we want to keep patients as safe as possible). Deep learning is especially powerful here because it can identify complex noise patterns in CT images and remove them, allowing us to achieve high image quality at even lower radiation doses.
The second area is medical image analysis, where we develop imaging algorithms to automatically segment structures in the heart and extract quantitative measurements that clinicians can use. For example, we can use machine learning to identify and quantify tissue patterns in the heart that are associated with disease.
The third area is computational cardiac mechanics: taking those images and building patient-specific models to quantify how the heart moves and functions, which can serve as important biomarkers for conditions like heart failure.
Engineering and health
Learn more about the ME biomechanics research that bridges boundaries between engineers and medical professionals.
Originally published April 13, 2026